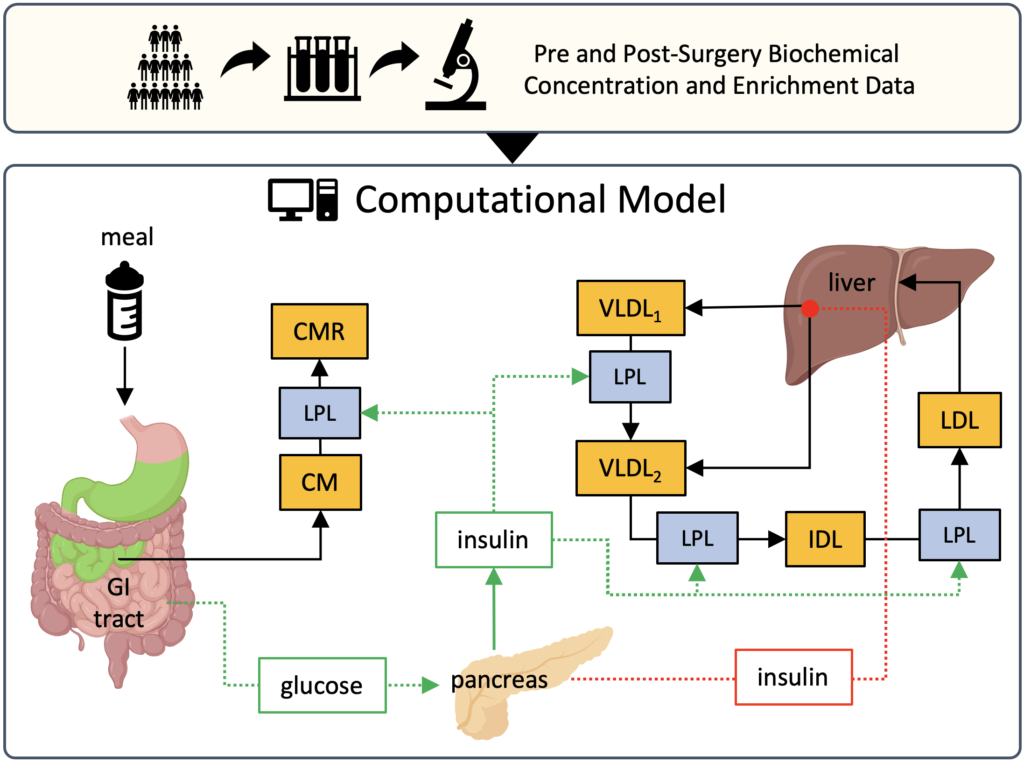
Since lipids are water-insoluble molecules, they are transported from the intestines and liver to the peripheral tissues via lipoproteins. In this project, our main goal is to develop physiology-based computational models of human lipoprotein metabolism by utilizing clinical data to investigate factors that regulate this metabolism, and to understand the effects of bariatric surgery. The novel computational models, which will be developed during the project, are aimed to guide clinical research and improve the efficiency of bariatric surgeries.
Besides significant weight loss, bariatric surgeries induce remarkable metabolic improvements in energy metabolism. After surgery, significant improvements take place in lipoprotein metabolism, insulin sensitivity, plasma glucose, and plasma lipid profiles of patients. Although all these improvements are well recognized, due to the complex nature of energy metabolism, cause and effect relation between them are not fully understood. Our goal is to investigate these complex interactions by developing physiology-based mathematical models of human lipoprotein metabolism that utilize data collected from patients that went through bariatric surgery.
Most of the existing lipoprotein metabolism models in the literature are steady-state pre-prandial hepatic models. Therefore, they fail in incorporating regulatory factors like insulin and explaining postprandial dynamics. Furthermore, existing models don’t integrate hepatic and digestive systems comprehensively, and hence they cannot simulate the anatomical changes induced by bariatric surgery and their effects. With this project, we aim to overcome this limitation by developing novel computational models of hepatic and intestinal lipoprotein metabolisms comprehensively.
This project is funded by the Scientific and Technological Research Council of Turkey (TÜBİTAK) under grant number 119F232.
PUBLICATIONS
Yildirim, V., Ter Horst, K. W., Gilijamse, P. W., van Harskamp, D., Schierbeek, H., Jansen, H., Schimmel, A. W., Nieuwdorp, M., Groen, A. K., Serlie, M. J., van Riel, N. A., & Dallinga-Thie, G. M. (2023). Bariatric surgery improves postprandial VLDL kinetics and restores insulin-mediated regulation of hepatic VLDL production. JCI insight, 8(16), e166905. https://doi.org/10.1172/jci.insight.166905
Yildirim V, Sheraton VM, Brands R, Crielaard L, Quax R, van Riel NAW, Stronks K, Nicolaou M, Sloot PMA. A data-driven computational model for obesity-driven diabetes onset and remission through weight loss. iScience. 2023 Oct 23;26(11):108324. doi: 10.1016/j.isci.2023.108324. PMID: 38026205; PMCID: PMC10665812.