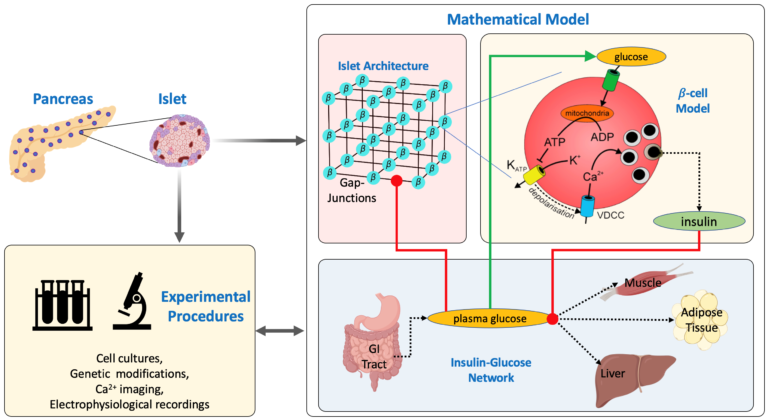
Insulin plays a key role in regulating energy metabolism and keeping plasma glucose concentrations under control. Insulin is secreted from pancreatic b-cell located on the islets of Langerhans in response to elevated plasma glucose levels in an oscillatory fashion with periods ranging from 2-10 minutes. Studies show that insulin oscillations increase the effectiveness of insulin, and insulin oscillations are lost in diabetic patients and their close relatives. Although the association between impaired insulin oscillations and diabetes is evident, the cause-consequence relation between them isn’t fully understood.
Insulin oscillations result from periodic electrical activity generated by pancreatic b-cells and their intra and inter-islet synchronization. The mechanism for inter-islet synchronization is not fully understood but intra-islet synchronization result from the gap-junctional connections between neighboring b-cells. Gap-junctional channels on pancreatic b-cells comprises primarily connexin36 proteins (cx36). The expression of cx36 protein is regulated by several factors including glucose. In this project, we investigate the bidirectional association between cx36 expression and the progression of diabetes via mathematical modeling and experimental procedures. In our lab, we develop physiologically based mathematical models of b-cell electrophysiology and insulin-glucose network to understand this bidirectional relation and test our model predictions in the lab.
This project is funded by the Scientific and Technological Research Council of Turkey (TÜBİTAK) under grant number 121F278.
PUBLICATIONS
An M, Akyuz M, Capik O, Yalcin C, Bertram R, Karatas EA, Karatas OF, Yildirim V. Gain of Function Mutation in K(ATP) Channels and Resulting Upregulation of Coupling Conductance are Partners in Crime in the Impairment of Ca2+ Oscillations in Pancreatic ß-cells. Mathematical Biosciences (2024): 109224. DOI: 10.1016/j.mbs.2024.109224, PMID: 38821258
Yildirim V, Sloot PMA. Protocol for analyzing emergence dynamics of diabetes with obesity using numerical continuation and bifurcation analysis. STAR Protoc. 2024 Mar 15;5(1):102880. doi: 10.1016/j.xpro.2024.102880. Epub 2024 Feb 12. PMID: 38349789; PMCID: PMC10876977.
Yildirim V, Sheraton VM, Brands R, Crielaard L, Quax R, van Riel NAW, Stronks K, Nicolaou M, Sloot PMA. A data-driven computational model for obesity-driven diabetes onset and remission through weight loss. iScience. 2023 Oct 23;26(11):108324. doi: 10.1016/j.isci.2023.108324. PMID: 38026205; PMCID: PMC10665812.